 It has been found that this reaction must be performed in either a polar media such as pyridine or methanol, or in a basic aqueous two-phase system. Upon heating (generally 60 C or higher), the tosylate anion will dissociate, generating the diazo compound 3. #Bamford stevens reaction pdf series#Variations and Improvements Initially, the decomposition of tosylhydrazones with base (and variations thereof) was used as a preparative procedure to synthesize a series of aryldiazomethanes and is still a standard method for their This procedure initially involves deprotonation of the tosylhydrazone 1 to form the corresponding anion. Bamford Stevens Reaction Series Of Aryldiazomethanes The migration of hydrogen may immediately follow, or be simultaneous with, the loss of dinitrogen. This pathway does not occur in the absence of protons, and in aprotic media, dinitrogen loss results in the formation of carbene intermediates 8. Miscellaneous Homologation Reactions 643 1 2 3 The mechanism for alkene formation was found to be dependent upon the reaction condition.įor instance, in protic media, the diazo compound 3 can be protonated to form a diazonium ion 5, resulting in the formation of a carbocation 6 upon loss of dinitrogen. The first step of the Bamford-Stevens reaction is the formation of the diazo compound 3, via the tosylate salt 2.6 Chapter 3. Mechanism The mechanism was originally proposed by Powell and Whiting,4 with krther corroboration by others soon thereafter. This valuable transformation has received extensive attention, as testified by the references cited herein. It is closely related to the Shapiro reaction, in which tosylhydrazones are treated with alkyl lithium reagents to form a lkene. Historical Perspectiw The titled reaction was first reported by William Randall Bamford and Thomas Stevens Stevens in 1952 at The University of Sheffield. Name Reactions for Homologations-2 3.1 Bamford-Stevens reaction Paul Humphries 3.1.1 Description 1 - 3 The Bamford-Stevens reaction is the formation of an alkene 4 by the treatment of the tosylhydrazone 1, of an aldehyde or a ketone, with a base.Ī few diazo compounds 3 can be isolated if mild temperatures are employed however in the majority of cases, the diazo compounds thermally decompose to form alkenes. 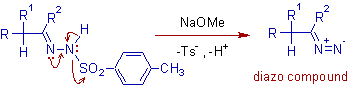
#Bamford stevens reaction pdf pdf#
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
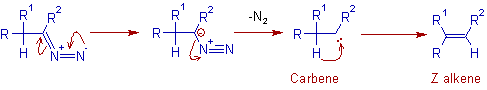
 RSS Feed
RSS Feed
